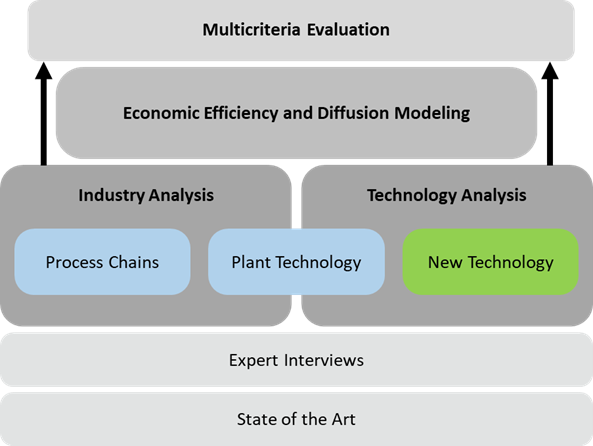
Cómo domar a tu dragón
Cuando una carga se atasca durante el temple, cada segundo cuenta y las decisiones improvisadas pueden aumentar el riesgo. En esta entrega de “Martes Técnico“, Bruno Scomazzon, gerente general de Precision Heat Treat Ltd., describe un procedimiento de respuesta de emergencia paso a paso para este escenario, uno de los más peligrosos en el tratamiento térmico con atmósfera. Basándose en la experiencia real, esta guía tiene como objetivo ayudar a las empresas a desarrollar sus propios procedimientos eficaces para mantener la seguridad, controlar las condiciones del horno y coordinarse con los servicios de emergencia en situaciones de alto riesgo.
Este artículo informativo se publicó por primera vez en Ingles en Heat Treat Today’s February 2026 Annual Air & Atmosphere Heat Treating print edition.

Si tiene comentarios o preguntas sobre este artículo, háganoslo saber en: editor@heattreattoday.com.
Traducido por Víctor Zacarías. To read this article in English, click here.
Descripción del escenario
Se ha transferido una carga a la cámara de temple y el elevador está descendiendo al aceite, pero la carga se atasca y no se sumerge por completo. La puerta interior se cierra correctamente, y la puerta exterior (frontal) permanece cerrada.
Esta es una situación de altísimo riesgo que requiere el estricto cumplimiento de los procedimientos de emergencia. El objetivo es proteger: primero al personal (minimizar la posibilidad de lesiones o que la situación empeore), luego las instalaciones y, finalmente, el equipo.
1. Medidas inmediatas
NO abra la puerta exterior
Es posible que sienta la tentación de evaluar la situación, pero resista la tentación. NO se coloque frente a la puerta exterior ni justo al lado de ella, y nunca la abra mientras la carga esté “colgada”. Abrir esta puerta puede introducir oxígeno en una cámara caliente, lo que provocaría:
- Explosiones o incendios repentinos (flash fire).
- Pérdida de contención debido a deformación de la puerta o falla mecánica.
En casos extremos, la puerta exterior puede resultar dañada (arrancada, atascada o parcialmente abierta) y presentar llamas visibles. Esto requiere alertar inmediatamente a los bomberos.
Si la puerta exterior no se puede cerrar
En este caso, notifique inmediatamente a los bomberos e indíqueles que se preparen para una respuesta con espuma. NO permita el uso de agua. ¡Esto podría provocar reacciones violentas con el aceite o la atmósfera y propagar el fuego!
El personal de respuesta capacitado debe:
- Colocarse el equipo de protección personal (EPP).
- Preparar el equipo de extinción de incendios.
- Estar listos para proteger los sistemas críticos hasta que lleguen los bomberos.
NO apague el horno.


2. Mantener el suministro eléctrico
Para garantizar que los sistemas esenciales permanezcan activos, debe mantener el suministro eléctrico. Asegúrese de que los siguientes sistemas permanezcan activos:
- Cambie el modo del horno de automático a manual. Esto evitará cualquier secuenciación del PLC que active automáticamente las puertas, los elevadores y los manipuladores.
- Mantenga las llamas piloto encendidas.
- Mantenga el enfriamiento del aceite en funcionamiento para evitar el sobrecalentamiento del tanque.
- Apague los calentadores de aceite para evitar una carga térmica adicional en el tanque de temple.
- Mantenga la agitación del temple a baja velocidad durante todo el proceso para ayudar a disminuir la temperatura en la superficie de interfaz entre la carga y el aceite. Esto evita la estratificación y disipa el calor por radiación en el aceite.
- Mantenga el recirculador en funcionamiento.
- Mantenga la instrumentación en funcionamiento para el monitoreo.
NOTA: La pérdida de estos sistemas elimina la visibilidad, el control de la atmósfera y las opciones de respuesta seguras.
3. Gestión de la atmósfera
Mantenga una atmósfera protectora y una presión positiva en el horno para evitar la entrada de oxígeno y la combustión incontrolada:
- Ajuste el control del potencial de carbono a “0”.
- Cierre el suministro de gas de enriquecimiento.
- Cierre el suministro de amoníaco.
- Cierre el suministro de aire de dilución.
Purga de nitrógeno
Estos pasos dependen de si se dispone de una purga de nitrógeno; se recomienda encarecidamente que esté disponible para todas las unidades con temple integral o de paso directo. Asegúrese de comprender cuánto tiempo tarda su horno en purgar completamente el gas endotérmico. Si bien la norma NFPA 86 recomienda cinco ciclos de purga, algunos expertos aconsejan prever hasta diez por hora en caso de emergencia. Cada horno debe contar con datos de purga establecidos en condiciones normales para que los operadores puedan actuar con confianza cuando el tiempo es crucial.

- Inicie inmediatamente una purga de nitrógeno (si está disponible) y manténgala durante todo el evento.
- Utilice al menos el flujo mínimo especificado en su documentación. Si es seguro, se puede utilizar un flujo mayor para ayudar a desplazar los gases inflamables de las cámaras de calentamiento y temple.
- Mantenga la temperatura del horno a 815°C (1500°F) durante la purga.
Pueden quedar espacios residuales de gas Endo atrapados en zonas con poca ventilación. Si la temperatura de la cámara desciende por debajo del punto de ignición antes de que se haya desplazado todo el gas inflamable, la entrada de oxígeno podría provocar una explosión. En algunos casos, el Endo atrapado y los desequilibrios de presión pueden causar fugas repentinas, en las que se expulsa aceite o gas debido a la acumulación de presión interna.
Después de la purga
El objetivo de la purga con nitrógeno es desplazar el gas endotérmico con una atmósfera inerte, manteniendo una temperatura elevada para facilitar la combustión de los gases inflamables residuales y prevenir la formación de mezclas peligrosas. Este proceso debe garantizar una presión positiva en todo el horno.
- Una purga seguida de enfriamiento por inmersión de nitrógeno es un método válido si la purga se ha completado de forma verificable.
- Según el tamaño del horno y la velocidad de enfriamiento:
- Los hornos más grandes pueden enfriarse lo suficientemente lento como para completar la purga.
- Las unidades más pequeñas o de enfriamiento más rápido pueden requerir un breve mantenimiento de la temperatura antes del enfriamiento controlado o el enfriamiento por inmersión.
NOTA: Una vez que la carga suspendida se enfríe a una temperatura segura (aproximadamente 65°C), realice el apagado estándar del equipo.
Sin nitrógeno (en Endo)
Si no hay purga de nitrógeno, o esta es insuficiente, la única opción es dejar que la carga acumulada se enfríe en el vestíbulo mientras se continúa quemando Endo y se mantiene la temperatura del horno a 1500°F. Una vez que el vestíbulo/tanque de aceite se enfríe por debajo de 150°F y haya pasado el peligro, inicie el apagado estándar del horno.
4. Gestión de la seguridad
- Alerte inmediatamente al cuerpo de bomberos local. Si la situación se vuelve incontrolable o si existe alguna duda sobre la capacidad de mantener el control, evacúe las instalaciones y espere a que lleguen profesionales capacitados. La seguridad del personal de la planta es primordial.
- Notifique al departamento de seguridad de la planta y a la administración del sitio.
- Evacúe a todo el personal no esencial del área de tratamiento térmico.
- Informe a todos los departamentos que se está produciendo un incidente de alto riesgo.
Los bomberos son más eficaces cuando conocen sus instalaciones antes de que ocurra una emergencia. Asegúrese de que conozcan la distribución de sus operaciones, incluyendo:
- Ubicación y tamaño de los tanques de aceite
- Paneles eléctricos
- Válvulas de cierre de gas
- Zonas calientes
5. Periodo de enfriamiento controlado
- Mantenga la protección con atmósfera durante todo el evento.
- NO abra las puertas hasta que la temperatura del vestíbulo sea baja y estable.
- El tiempo de enfriamiento dependerá de la masa de la carga y la retención de calor. Prevea cinco horas o más.
- Utilice la estabilidad de la presión del horno, las observaciones de los efluentes y el comportamiento de los gases como indicadores indirectos de la temperatura.
6. Procedimiento de recuperación de la carga
- Una vez enfriado y estabilizado, realice el apagado estándar, comenzando con la eliminación del gas endotérmico, si corresponde.
- NO intente retirar la carga manualmente hasta que el sistema esté verificado como seguro.
- Solo el personal de mantenimiento puede recuperar la carga, utilizando equipo de protección personal (EPP) y las herramientas adecuadas.
7. Familiarización con el cuerpo de bomberos
Toda instalación debe establecer una buena relación con el cuerpo de bomberos local antes de que ocurra una emergencia. Procure revisiones anuales e identifique lo siguiente:
- Número de hornos
- Volumen de los tanques de aceite de temple para extinción de incendios
- Ubicación de las zonas calientes y los paneles de control
- Puntos de parada de emergencia
Las puertas atascadas suelen deberse a fallos en las válvulas neumáticas. Cerrar el suministro de aire comprimido y purgarlo puede permitir que el mecanismo se reinicie. Consulte siempre el manual del equipo o al fabricante antes de intentar cualquier solución.
Considere que el inspector de bomberos que realice las revisiones no es necesariamente quien acudirá a combatir los incendios; capacite a quienes sí lo harán.
Protocolo posterior al incidente
Antes de volver a poner en funcionamiento el horno, asegúrese de:
- Realizar una investigación formal.
- Identificar y corregir la(s) causa(s) raíz.
- Documentar todos los parámetros clave y las acciones tomadas.
- Capacitar nuevamente a los operadores según sea necesario.
Señalización del horno
Es probable que un operador lea el plan de seguridad, pero podría olvidar un protocolo vital durante una emergencia. Contar con advertencias claras y llamativas, impresas y colocadas en el panel, que el operador pueda retirar y utilizar en caso de emergencia, puede ser de gran utilidad.
Reflexiones finales
No podemos predecir todas las consecuencias. Ningún procedimiento puede contemplar todas las variables posibles en una emergencia real. Una vez que un evento se pone en marcha, lo único que podemos hacer es responder con el mejor criterio, capacitación e intenciones, priorizando siempre la seguridad de las personas.
Este documento pretende ser una referencia práctica: una guía estructurada elaborada con esmero, experiencia real y buenas prácticas. No es una solución universal, sino una herramienta para ayudar a los equipos a crear o mejorar sus propios procedimientos eficaces y a responder de forma adaptativa en situaciones de alto riesgo.
La preparación contra incendios es esencial en toda planta de tratamiento térmico. Los incendios ocurren, y no siempre son pequeños. Es fundamental saber cuándo actuar, cuándo evacuar y cuándo pedir ayuda. Los manuales de equipos proporcionan una base, pero la preparación mediante capacitación y planificación es la mejor defensa.
Agradecimientos: El autor agradecer a Daniel H. Herring, “The Heat Treat Doctor,” a The HERRING GROUP, Inc., y a Avery Bell de Service Heat Treat en Milwaukee por sus valiosas contribuciones.
Acerca del autor:

Gerente General
Precision Heat Treat Ltd.
Bruno Scomazzon es el gerente general de Precision Heat Treat Ltd. en Surrey, Columbia Británica, Canadá, y cuenta con más de 40 años de experiencia en procesos metalúrgicos y operaciones de tratamiento térmico.
Para más información: Contacte a Bruno en bruno@precisionheattreat.com.

Cómo domar a tu dragón Read More »