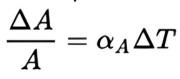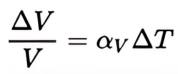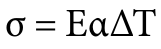Ask The Heat Treat Doctor®: What Are the Differences Between Intergranular Oxidation (IGO) and Intergranular Attack (IGA)?

The Heat Treat Doctor® has returned to offer sage advice to Heat Treat Today readers and to answer your questions about heat treating, brazing, sintering, and other types of thermal treatments as well as questions on metallurgy, equipment, and process-related issues.
Today’s Technical Tuesday is a pre-release foretaste of the great content you can find in Heat Treat Today’s July 2025 Super Brands print edition.
Heat treaters and metallurgists speak a language unique to our industry and it can be confusing at times; terms like intergranular oxidation (IGO) and intergranular attack (IGA) are good examples, as these terms are often (incorrectly) used interchangeably. While these two phenomena sound similar, they have distinct mechanisms, causes, and impacts on material properties. Expert Dan Herring explores them more below.
What is IGO?

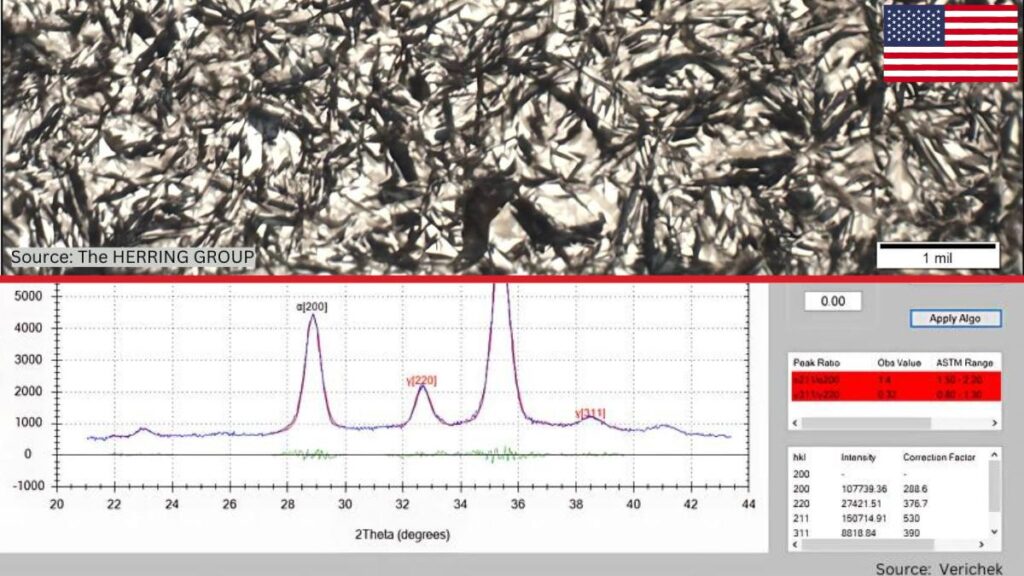
IGO is a process by which oxygen preferentially reacts with the metal surface at the grain boundaries, creating oxides (Figure 1). It is not uncommon, for example, to see IGO present after a hardening process run in an Endothermic or nitrogen/methanol atmosphere. As parts are heated to austenitizing temperature, oxygen present due to minute air leaks or produced during the various chemical reactions with the atmosphere results in IGO. The grain boundaries are highly susceptible to oxidation because these are areas where different crystallographic grains meet and are areas of high energy due to the atomic mismatch and disruption of the regular crystal lattice structure.

Source: The HERRING GROUP, Inc.
What is IGA?

Source: The HERRING GROUP, Inc.
IGA, on the other hand, is a broader term that refers to a corrosion phenomenon (aka chemical attack) that specifically targets the grain boundaries of a material. Unlike IGO, intergranular attack (Figure 2) is not limited to oxidation reactions but encompasses a variety of forms of attack involving such things as the formation of precipitates, the dissolution of material at grain boundaries, or the creation of corrosion cracks. Common forms of IGA include stress corrosion cracking (SCC) or sensitization in stainless steel.
In stainless steels, IGA is often triggered by high-temperature environments, usually in the range of 840º – 1560ºF (450 – 850°C) where carbon reacts with chromium to form chromium carbides at the grain boundaries, thus reducing the material’s resistance to corrosion in localized regions. In other alloys, factors like pH, chloride concentration, and temperature can lead to IGA.
Both IGO and IGA weaken the material’s structural integrity or lead to embrittlement compromising the material’s integrity.
Effect on Material Properties
The main effect of intergranular oxidation is the degradation of the mechanical properties, particularly a reduction in both ductility and toughness. As oxidation progresses along the grain boundaries, the material tends to become brittle, which can lead to premature failure under certain types of stress or thermal cycling. IGO often appears visually as a uniform discoloration or thin oxide layer on the surface. Surface pitting is not typically observed.
By contrast, IGA often appears as visible cracks, pits, or localized regions where the metal has been attacked (along the grain boundaries). This leads to a reduction in mechanical strength and can lead to SCC under certain circumstances. IGA can severely compromise the integrity of the material, particularly in critical applications like pipelines, pressure vessels, and nuclear reactors.
Materials Involved
IGO is most commonly observed in steel, aluminum, titanium, and nickel-based alloys, not only during heat treatment but when exposed to oxidizing environments in high-temperature applications, which also result in degradation and loss of material strength and other properties.
IGA tends to be more prevalent in stainless steels, corrosion-resistant alloys, and aluminum alloys. It is especially noticeable in alloys that are susceptible to sensitization (where chromium carbides precipitate at grain boundaries), leading to localized corrosion and cracks. Alloys that form a passivating oxide layer can be more susceptible to IGA if that layer is disrupted.
Principal Concerns
The main concern with intergranular oxidation is material embrittlement, leading to reduced ductility and potential failure under mechanical stress, especially in high-temperature applications. It can also affect the integrity of critical components, such as those used in aerospace or power generation industries.
By contrast, the primary impact of intergranular attack is loss of material strength, leading to structural failure, often without any clear outward signs (e.g., under chloride-induced SCC). It is more likely to cause immediate failure or a dramatic loss in performance, especially in structures exposed to corrosive environments.
How to Detect IGO and IGA
IGO is typically detected by examining the material’s surface using optical or scanning electron microscopy (SEM). Non-destructive techniques, such as X-ray diffraction (XRD), can also be used.
IGA is usually detected through methods like microstructural examination, electrochemical testing, or failure analysis. Techniques, such as SEM or energy-dispersive X-ray spectroscopy (EDS), can be used to examine the grain boundary regions for signs of corrosion.
How to Avoid IGO and IGA
IGO can be avoided by one or more of the following:
- Environmental control: Making sure the heat treat furnace has no leaks, reducing oxygen partial pressure or controlling the furnace atmosphere in high-temperature heat treat operations.
- Alloy design: The use of materials with stable oxide-forming elements (e.g., chromium, titanium and aluminum) or alloys with high resistance to oxidation (e.g., nickel-based superalloys).
- Temperature control: Maintaining lower process temperatures and shorter times where possible to prevent oxidation at the grain boundaries.
- Coatings and surface treatments: Application of protective coatings, such as copper plating, post-heat treatment aluminizing, or chrome plating, to reduce oxygen interaction with the grain boundaries during service.
IGA can be avoided by one or more of the following:
- Environmental control: Reducing exposure to aggressive chemicals (e.g., chloride ions) by maintaining proper pH levels or using inhibitors in post-cleaning processes.
- Proper alloy selection: Selecting materials resistant to intergranular corrosion (e.g., low carbon “L” grades of stainless steel or alloys with improved grain boundary stability).
- Heat treatment: Avoiding sensitization of stainless steel by proper heat treatment methods that prevent the formation of chromium carbides at grain boundaries.
- Stress relief: Reducing the likelihood of stress corrosion cracking by managing internal stresses during manufacturing and in-service conditions.
Key Differences
The differences between these phenomena are summarized in Table 1.

Source: The HERRING GROUP, Inc.
Summing Up
While both IGO and IGA involve attack at the grain boundaries, they differ in their mechanisms, causes, and effects. From a heat treater’s perspective, IGO most often results at high temperature in oxygen-bearing furnace atmospheres, while IGA often results from pre- or post-heat treatment processing (cleaning, passivation, plating, etc.). Proper material selection, furnace and environmental control, awareness of what can happen, and inspection for these effects are key to preventing them from occurring.
References
Roberge, Pierre R., Corrosion Engineering: Principles and Practice, Mc-Graw Hill LLC, 2008.
Stene, Einar S., Fundamentals of Corrosion: Mechanisms, Causes, and Monitoring.
Schweitzer, Philip A., Fundamentals of Corrosion: Mechanisms, Causes and Preventative Methods, CRC Press, 2009.
About the Author

“The Heat Treat Doctor”
The HERRING GROUP, Inc.
Dan Herring has been in the industry for over 50 years and has gained vast experience in fields that include materials science, engineering, metallurgy, new product research, and many other areas. He is the author of six books and over 700 technical articles.
For more information: Contact Dan Herring at dherring@heat-treat-doctor.com.
For more information about Dan’s books: see his page at the Heat Treat Store.