
Ask The Heat Treat Doctor® has returned to bring sage advice to Heat Treat Today readers and to answer your questions about heat treating, brazing, sintering, and other types of thermal treatments as well as questions on metallurgy, equipment, and process-related issues. In this installment, Dan Herring discusses the science behind pH — what it really measures, and why it matters — and offers practical guidance on monitoring water quality in open and closed systems found throughout the heat treat shop.
This informative piece was first released in Heat Treat Today’s May 2026 Sustainable Heat Treat Technologies print edition.
Introduction to pH
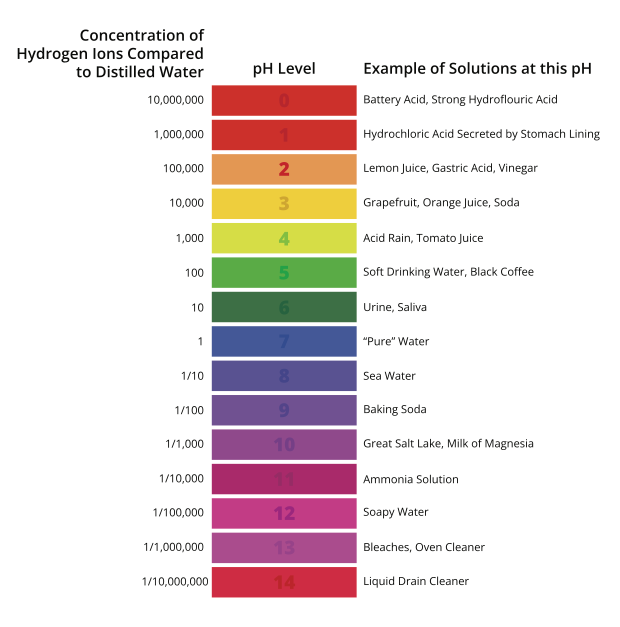
The term “pH” is used to describe a unit of measure that indicates the degree of acidity or alkalinity of a solution. It is measured on a scale of 0 to 14 (Table A). pH is an abbreviation that stands for the “potential of hydrogen”; the “p” being the symbol for potential (or power) and “H” the symbol for hydrogen.
A Slightly Deeper Dive
What most people don’t realize is that pH is a complex concept rooted in chemical equilibrium, thermodynamics, and electrochemistry. The formal definition of pH is “the negative logarithm of the hydrogen ion activity” and can be expressed mathematically by the following formula where au+ is the activity of hydrogen ions, a dimensionless quantity (Rumble 2024):
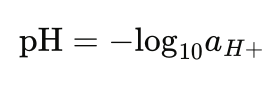
In this form, pH provides a way of expressing the degree of the activity of an acid or base in terms of its hydrogen ion activity. Acids and bases have, respectively, free hydrogen [H+] and free hydroxyl [OH−] ions. Since the relationship between hydrogen ions and hydroxyl ions in a given solution is constant for a given set of conditions, either one can determine the other. In other words, pH is really a measurement of both acidity and alkalinity, even though by definition it is a selective measurement of hydrogen ion activity.
Since pH is a logarithmic function, a change of one pH unit represents a tenfold change in hydrogen ion concentration, that is, of both the hydrogen ion and the hydroxyl ion at different pH values (Table A). Note that each decrease in pH by one pH unit means a tenfold increase in the concentration of hydrogen ions.
A Little Chemistry
In school, we learned that all substances are made up of millions of tiny atoms. These atoms combine to form molecules. In water, for example, each molecule is made up of two hydrogen (H) atoms and one oxygen (O) atom. The formula for a molecule of water is expressed by the familiar symbol H2O. That is, there are two hydrogen atoms needed for each oxygen atom to form a stable compound.
Now, the behavior of pH in aqueous systems is governed by the equilibrium of water to form positive and negative ions (so-called self-ionization), which can be expressed as:
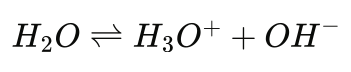
or in the following form we more commonly think of:
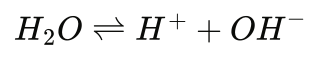
Hency, at 25°C (Kw = 1.0 x 10
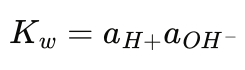
Then for pure water, where aH+ = a0H-, we have that aH+ = 10-7 hence pH = -log10 (10-7) = 7.00 which is neutrality at 25°C (77°F).
Finally, it is important to note that Kw is temperature-dependent: it increases with temperature, meaning neutral pH decreases slightly as temperature rises (e.g., ~6.14 at 100°C). Therefore, “neutral pH” is not always 7 — it depends on thermal conditions.
A Practical Application — Water Quality in the Heat Treat Shop
Water is used in most of our heat treat shops for a variety of purposes, perhaps less than before but still vitally important. Examples include parts washers, heat exchangers, water cooled bearings on fans and rolls, seals on pit furnace covers, water cooled jackets on continuous furnaces, water cooled jackets for quench tanks, top or side cooling chambers, inner doors and plate coils, and make up water for water systems, to name a few.
Water quality requirements are often defined differently for open systems (Table B) and closed (recirculated) systems (Table C). Open systems are typically more problematic as the issue of water quality varies. Water is often classified as “soft” or “hard” depending on its mineral content (i.e., the amount of calcium and magnesium dissolved in the water). Soft water has an ideal hardness of approximately 120 ppm (7 grains/gallon). Hard water often results in the formation of mineral deposits, which can lead to blockages in water systems (Figure 1).

Furthermore, we must ensure that the water being discharged from our heat treatment operations is clean and meets EPA standards. Finally, we must be especially careful to avoid cross-contamination from other sources in the shop (e.g., polymers, quench oils, chemicals).
In Summary
Two little consonants, pH, are deceptively simple yet so profoundly important. They represent the thermodynamic state of solutions, but in reality, link microscopic interactions with real world issues. As heat treaters, our focus is to not take our water supply and water systems for granted since unexpected surprises, unwanted downtime, and expensive repairs can result. When is the last time you tested your water?
References
Herring, Daniel H. 2015a. Atmosphere Heat Treatment. Vol. 2. Southfield, MI: BNP Media.
Herring, Daniel H. 2015b. “The Importance of pH.” Industrial Heating, January.
Heatlink Group. 2006. Water Quality in Hydronic Systems. June 21, 2006. https://www.heatlink.com/sites/default/files/Info%20Sheet/L2329-Water-Quality-in-Hydronic-Systems-2006-06-21.pdf.
Decelles, P. 2002. The pH Scale. Johnson County Community College. Archived webpage. http://staff.jccc.net/pdecell/chemistry/phscale.html.
Rumble, John R., ed. 2024. CRC Handbook of Chemistry and Physics. 105th ed. Boca Raton, FL: CRC Press.
About the Author

“The Heat Treat Doctor”
The HERRING GROUP, Inc.
Dan Herring has been in the industry for over 50 years and has gained vast experience in fields that include materials science, engineering, metallurgy, new product research, and many other areas. He is the author of six books and over 700 technical articles.
For more information: Contact Dan at dherring@heat-treat-doctor.com.
For more information about Dan’s books: see his page at the Heat Treat Store.







