Jim Roberts of U.S. Ignition engages readers in a Combustion Corner editorial about the double-edged sword of heat recovery technology — explaining how efforts to reduce fuel consumption inadvertently drove up NOx emissions, and how flue gas recirculation (FGR) emerged as the design solution capable of cutting both fuel use and emissions by up to 50%.
This editorial was first released inHeat Treat Today’s February 2026 Annual Air & Atmosphere Heat Treating print edition.
A furnace guy walks into the heat treating plant and says to the operators standing nearby, “This exhaust system and these burners all have a negative attitude.” The other furnace guys say, “They better be negative, or they would not work well!” As if we don’t have enough negativity swirling around in our world as it is, now we are happy about it?
In the Annual People of Heat Treat (September 2025) we talked about the types of burners that were developed as heat treating and furnace sciences and combustion designs evolved. We also chatted about how the advent of new fuels and government regulations was going to take a chunk of our attention in the coming years — for example, pollution laws coming to the forefront of our industry in the late ‘70s and onwards. Interesting new burner designs sprung up, primarily, as you recall, to address the usage of gas. In other words, how can we reduce fuel usage?
But First, NOx
The cost of gas skyrocketed for a stretch and it led us first to energy reduction plans. But with heat recovery sciences came the phenomenon of higher flame temperatures. When you get higher flame temperatures, you can sometimes (okay… all the time) generate NOx. One of the primary constituents of atmospheric pollution is NOx, and it became a prime target for reduction by the EPA and other governing air quality folks. As it should be.
Just a quick step back to the “remind me again, Jim” world. What do we breathe? Air, right? We have to have oxygen. But what we tend to forget is that air is roughly 79% nitrogen. So, what we breathe is actually nitrogen spiked with oxygen, and the fuel that we generally burn, natural gas, has some nitrogen in it too.
Natural gas can have as much as 5% nitrogen in it, although membrane filtering usually controls pipeline gas content at around 1%. The point is that nitrogen is the dominant gas in our combustible portfolio, and when we make it really hot, it makes NOx. And that is considered bad for all of us. So, NOx from fuel-borne nitrogen can be released at temperatures as low as 1400°F. Sometimes that is referred to as “sudden NOx” because it releases quickly. All of us Furnace Guys know that 1400°F ain’t nothing in our world.
The second form of NOx is referred to as “thermal NOx” and that is the major source of NOx in our world. That is when we heat the air we are combusting in a burner, burning off most of the 21% oxygen. Then, flame temperature climbs, and continues to now superheat and try to burn that remaining 79% of nitrogen. As temperatures approach 2300°F, the magic happens.
Thermal NOx forms significantly at high combustion temperatures, typically starting above 1300°C (2372°F), with formation increasing exponentially as temperatures rise, especially above 2800°F (1538°C), due to atmospheric nitrogen and oxygen reacting at peak flame temperatures. Does anybody remember what happens to flame temperatures when we preheat the combustion air (recuperation, recirculation, etc.)? Flame temp and heat transfer increase and we go up to theoretical flame temperatures of 3200°F without even working at it.
Solving Energy Efficiency Through Design
So, let’s return to the original question: What happened when we tried to only save gas with heat recovery? Answer: We installed energy efficient burners but increased the emissions footprint in doing so. We cut down on energy expenditure but made exhaust an issue with the higher temps.
For most industrial and commercial applications, the optimal range for flue gas recirculation (FGR) is between 10% and 25% as this range offers significant NOx reduction without compromising combustion stability or efficiency. By adjusting the pressures coming into the burner and then balancing the exhaust outlet pressures over the heat exchanger body, normally with an extraction device called an “eductor,” we can dial in the percentage of recirculation the burners are operating under.
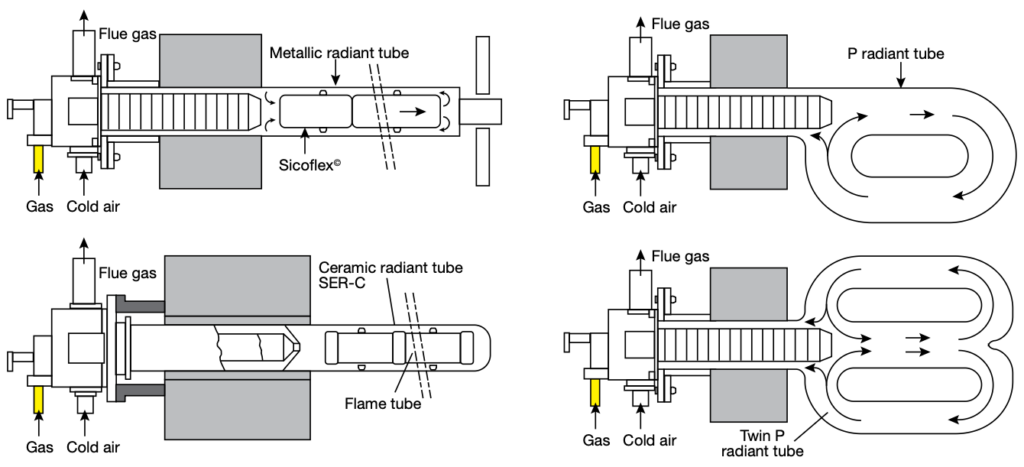
With this design, I have seen fuel and emission reductions of 50% when compared to the existing conventional combustion systems. It really is a testament to what design and research can produce for us (Figure 1).
We’ll look more closely at these designs next time.
About The Author:

President
US Ignition
Jim Roberts president at U.S. Ignition, began his 45-year career in the burner and heat recovery industry focused on heat treating specifically in 1979. He worked for and helped start up WB Combustion in Hales Corners, Wisconsin. In 1985 he joined Eclipse Engineering in Rockford, IL, specializing in heat treating-related combustion equipment/burners. Inducted into the American Gas Association’s Hall of Flame for service in training gas company field managers, Jim is a former president of MTI and has contributed to countless seminars on fuel reduction and combustion-related practices.
For more information: Contact Jim Roberts at jim@usignition.com.







